Calcium chloride molar mass4/13/2023 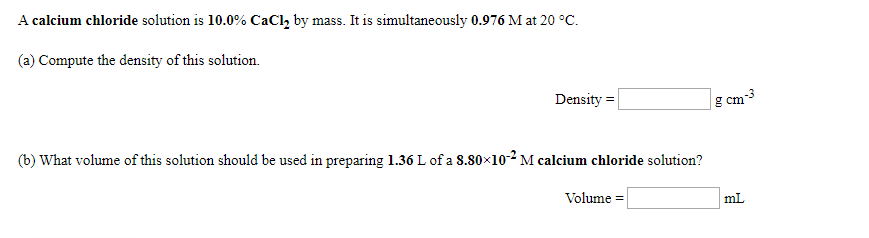
Calcium Chloride Dihydrate is having a melting point of 176C. On further evaporation it can be converted to Calcium Chloride Dihydrate Crystals or Calcium Chloride Fused. Commonly used to dry gases, the Calcium Chloride (Fused) made available by us is very cost effective.ĬaCl2 is available in Calcium Chloride Solution say of 30-35%. Our Calcium Chloride (Fused) is an anhydrous compound that is hygroscopic and rapidly absorbs water. It is a salt that is solid at room temperature, and it behaves as a typical ionic halide. Provides artificial water hardness that helps set paper dyesĬalcium Chloride (Fused) offered by us is a chemical compound that is crystalline, lumpy or flaky is usually white and is very soluble in water.Works with fatty acid soaps on recycled paper.Provides artificial water hardness for better drainage.Increases web strength of corrugating media.Retains moisture lowers freezing point of water. 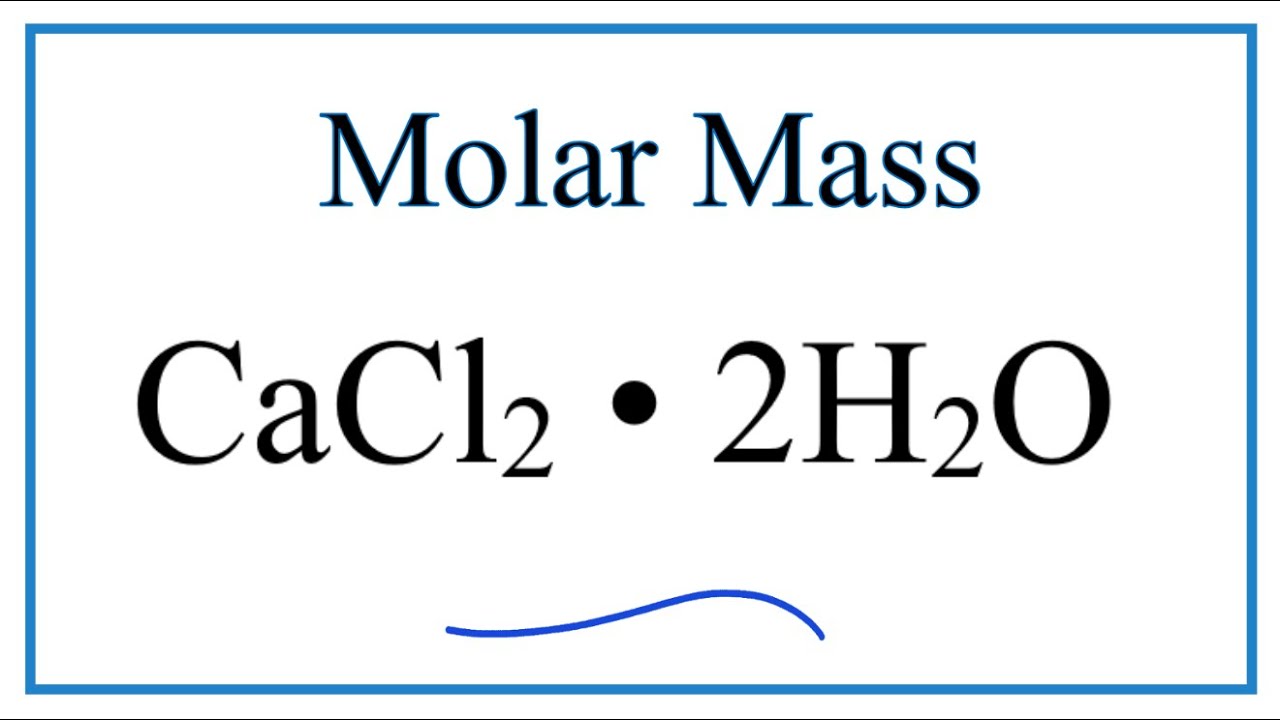
Dustproofing and freeze-resisting ore and coal.Removes Fluorides, Phosphates, Heavy Metals and oily wastes.Retains moisture which improves compaction of soil.Lowers freezing temperature of water put in tires for better traction.Solidifies loose, sandy soils when mixed with sodium silicate.Moisture absorbed from the air prevents dust formation.A dual credit course on the other hand is an official course at Loyola University Chicago. Paha777 Answer: ☆AP courses are part of the College Board organization that requires students to take a rigorous test at the end of the course to potentially earn college credit. How is the dual credit program different from the AP program. Therefore, a sample of calcium chloride contains twice as many chloride ions 2H2O: Calcium chloride dihydrate.A few things to consider when finding the molar mass for CaCl2 Ĭalcium Chloride is an ionic compound with the chemical formula CaCl2.Examples: Fe, Au, Co, Br, C, O, N, F.Įxplanation of how to find the molar mass of CaCl2 Use uppercase for the first character in the element and lowercase for the second character. It will calculate the total mass along with the elemental composition and mass of each element in the compound. Enter the molecular formula of the substance. This program determines the molecular mass of a substance. Recovery from the metal stress was considerable in the root fresh weight and almost completely in the root dry weight when 10.0 mM (1.9 EC), calcium chloride was supplied to the growing PB-1, the decrease in fresh and dry mass was higher with 1.0 mM Cd2+ than with the same level of Pb2+ and Cu2+. In the early growth phase of Sesamum indicum cv. Since the molar mass of Calcium is 40.08 g and the molSee more on Ĭalcium Chloride CaCl2 Molar Mass, Molecular Weight. Explanation: The molar mass of a compound is just the molar masses of every element in the compound added up. Q:What is the molar mass of calcium chloride ($$CaCl_2$$)?A:110.98 g. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together. ››More information on molar mass and molecular weight. Weights of atoms and isotopes are from NIST article. (1 u is equal to 1/12 the mass of one atom of carbon-12) Molar mass (molar weight) is the mass of one mole of a substance and is expressed in g/mol. Molecular mass (molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u). How many grams of CaCl2 (molar mass =111g/mol) are needed.Molar Mass / Molecular Weight of CaCl2.Calculate molar mass (calculate molecular weight, Molar.What is the molecular weight of this drug? – Get the answer to this question and access a vast question bank that is tailored for students.įormula unit mass = Atomic mass of C a + (2 × a t o m i c m a s s o f C l) = 4 0 + 7 1 = 1 1 1 Solve any question of Some Basic Concepts of Chemistry with:- Patterns of problems The molecular formula of calcium chloride is CaCl2. But if you look at the weight online, it says 110.98 g/”mol”, which is 2Ĭalculations: Formula: CaCl2 Molar Mass: 110.986 g/mol 1g=9.01014542374714E-03 mol Percent composition (by mass): Element Count Atom Mass %(by mass)ġ11 g/”mol” Weight of Ca = 40 Weight of Cl = 35.5 Molecular formula is CaCl_2 i.e. Calcium chloride cacl2 molar mass, molecular weight. convert grams cacl2 to moles or moles cacl2 to grams. this compound is also known as calcium chloride.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
